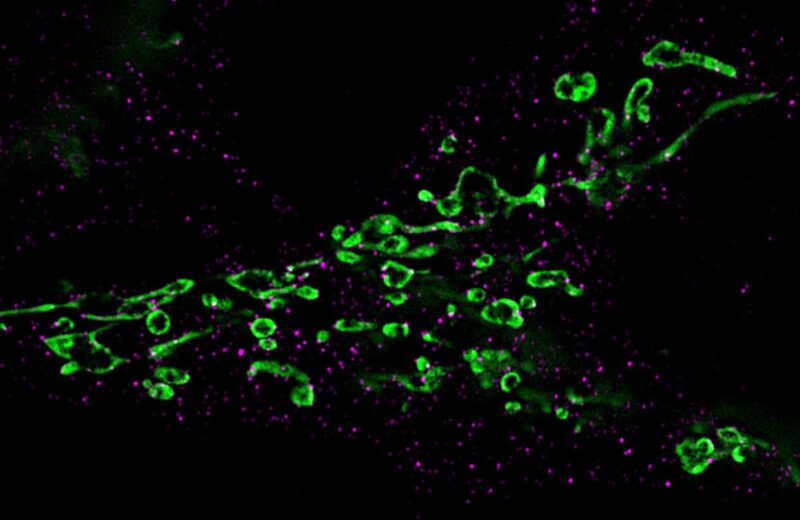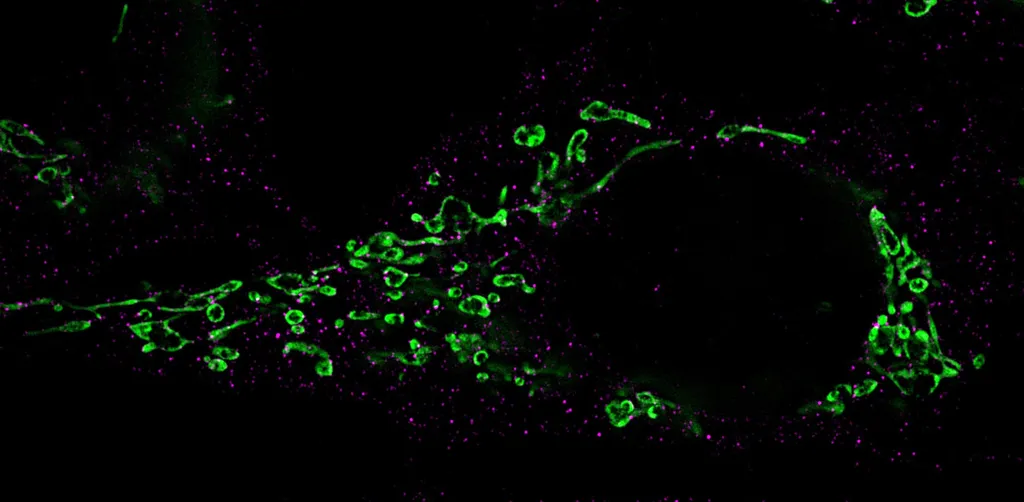
Fragmentation des mitochondries (vert) : Les protéines Drp-1 responsables de la désintégration sont marquées par des anticorps et colorées en magenta. Crédit : Chaire de Virologie / Université de Wuerzburg
Huit herpèsvirus différents sont connus à ce jour chez l’homme. Ils s’installent tous définitivement dans l’organisme après une infection aiguë. Dans certaines circonstances, ils se réveillent de cette phase de dormance, se multiplient et attaquent d’autres cellules. Cette réactivation est souvent associée à des symptômes, tels que les boutons de fièvre qui démangent ou le zona.
Au cours de l’évolution, la plupart des herpèsvirus ont appris à utiliser de petits RNA molecules, so-called microRNAs, to reprogram their host cells to their advantage. A research team led by Bhupesh Prusty and Lars Dölken from Julius-Maximilians-Universität (JMU) Würzburg in Bavaria, Germany, has now been able to show for the first time that a viral microRNA acts as a master regulator to induce the reactivation of the virus. In a study published today (May 4, 2022) the journal Nature, the researchers present the previously unknown cellular mechanism by which human herpesvirus 6 (HHV-6) triggers its own awakening.
Problems after reactivation of the virus
More than 90 percent of all people are infected with HHV-6 without noticing it. The virus probably only causes problems when it wakes up repeatedly.
Human herpesvirus 6 (HHV-6) is the common collective name for human betaherpesvirus 6A (HHV-6A) and human betaherpesvirus 6B (HHV-6B). HHV-6A has been described as more neurovirulent, and as such is more frequently found in patients with neuroinflammatory diseases such as multiple sclerosis. HHV-6 (and HHV-7) levels in the brain are also elevated in people with Alzheimer’s disease. HHV-6B primary infection is the cause of the common childhood illness exanthema subitum (also known as roseola infantum or sixth disease). It is passed on from child to child. Adults are unlikely to catch this disease since most people have had it by kindergarten, and once contracted, immunity develops, preventing future reinfection.
HHV-6 reactivation is suspected of impairing heart function, causing the rejection of transplanted organs, and triggering diseases such as multiple sclerosis or chronic fatigue syndrome (ME/CFS). In addition, recent studies suggest that this herpesvirus may be involved in the development of schizophrenia, bipolar disorder, and other diseases of the nervous system.
“How herpesviruses reactivate from a dormant state is the central question in herpesvirus research,” says JMU virologist Lars Dölken. “If we understand this, we know how to intervene therapeutically.” A previously unknown key to this is a viral microRNA called miR-aU14. It is the central switch that initiates the reactivation of HHV-6.
What the microRNA does in the cell
The regulatory miR-aU14 comes from the virus itself. As soon as it is expressed, it interferes with the metabolism of human microRNAs. In doing so, it selectively interferes with the maturation of several microRNAs of the miR-30 family. As a result, these important cellular microRNAs are no longer produced. This in turn affects a cellular signaling pathway, the so-called miR-30 / p53 / Drp1 axis.
Through this pathway, the viral miR-aU14 induces mitochondrial fragmentation. These cell structures are of central importance for energy production, but also for signal transmissions in the defense against viruses.
The viral miR-aU14 thus interferes with the production of type I interferons – messenger substances with which the cell signals the presence of viruses to the immune system. Because the interferons are missing, the herpesvirus is able to switch from a dormant to an active state undisturbed. Interestingly, the Würzburg research group was also able to show that the viral microRNA is not only essential for virus replication, but also directly triggers the reactivation of the virus from its dormant state.
How the research continues
The researchers now want to understand the exact mechanism by which the viral microRNA initiates the reactivation of the virus. In addition, there are first indications that other herpesviruses can also be reactivated via the same mechanism. This could reveal therapeutic options to either prevent reactivation of these viruses or to specifically trigger it in order to then eliminate the reactivating cells. Another goal is to understand the molecular consequences of mitochondrial fragmentation in detail.
For the first time, this work from Würzburg shows that a microRNA can directly regulate the maturation process of other microRNAs. This also opens up new therapeutic possibilities: Artificial small RNAs can be designed to specifically switch off individual members of microRNA families. Such subtle interventions were not possible until now.
Reference: “Selective inhibition of miRNA processing by a herpesvirus-encoded miRNA” by Thomas Hennig, Archana B. Prusty, Benedikt B. Kaufer, Adam W. Whisnant, Manivel Lodha, Antje Enders, Julius Thomas, Francesca Kasimir, Arnhild Grothey, Teresa Klein, Stefanie Herb, Christopher Jürges, Markus Sauer, Utz Fischer, Thomas Rudel, Gunter Meister, Florian Erhard, Lars Dölken and Bhupesh K. Prusty, 4 May 2022, Nature.
DOI: 10.1038/s41586-022-04667-4
Cooperation partners and sponsors
Several groups at JMU are conducting interdisciplinary research on this topic. They come from the Institute of Virology and Immunobiology, the Biocentres’ Chairs of Biochemistry, Biotechnology and Biophysics, and Microbiology, the Rudolf Virchow Centre and the Helmholtz Institute for RNA-based Infection Research. Researchers from the Free University of Berlin and the University of Regensburg were also involved.
The research was funded by the Helmholtz Institute for RNA-based Infection Research, the Solve ME/CFS Initiative (USA), the HHV-6 Foundation (USA), the Amar Foundation (USA) and by the European Research Council within the framework of an ERC grant.